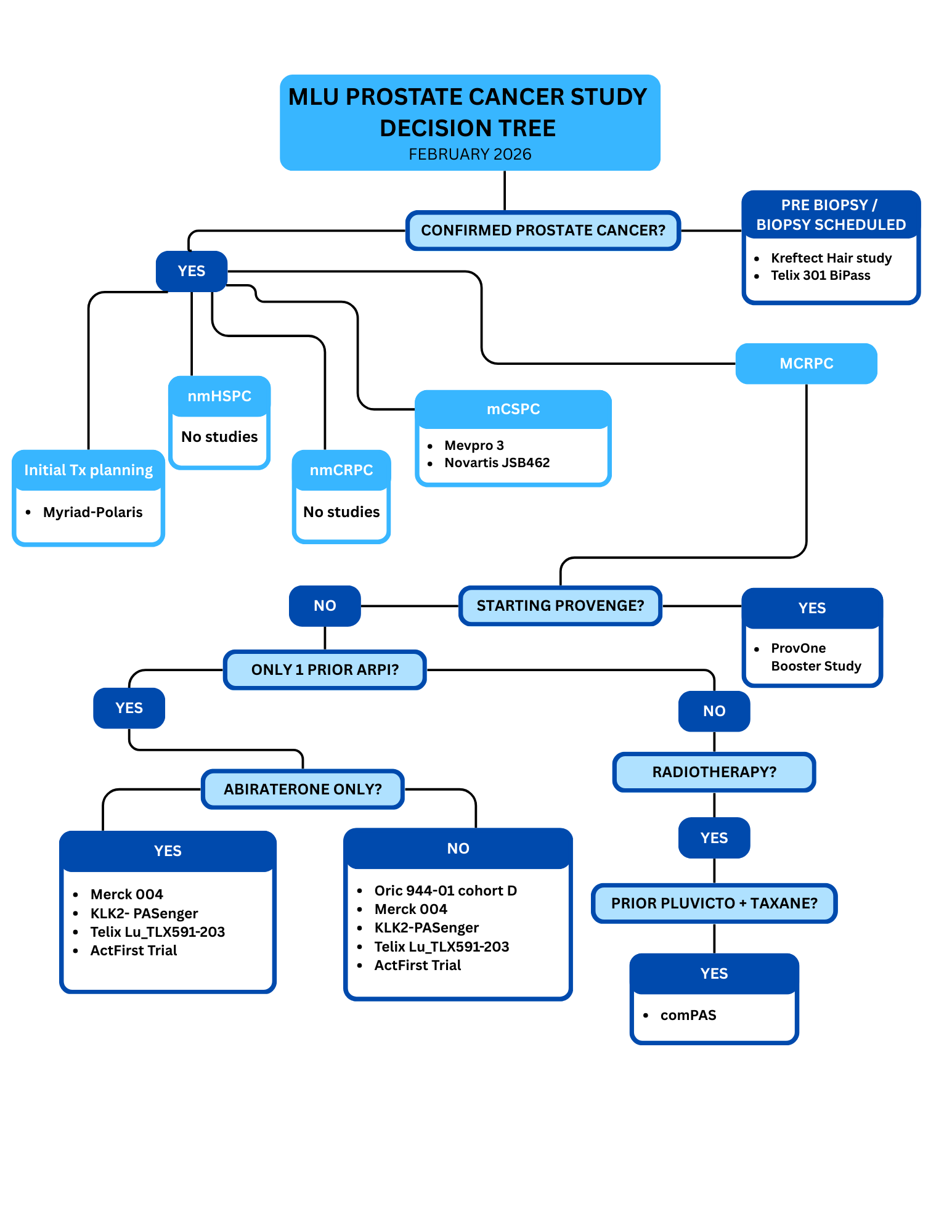
Prostate Cancer Study Decision Tree
Prostate Cancer Timeline
Pre-Diagnosis
Kreftect Hair Study
Telix 301 BiPass

Newly Diagnosed Treatment Planning

Active Surveillance

Primary Treatment

Biochemical Failure

mCSPC
- A phase 3, randomized, double-blind, placebo-controlled study of Mevrometostat (pf-06821497) with Enzalutamide in Metastatic Castration-Sensitive Prostate Cancer
- A Phase II, randomized, open-label, multi-center study of JSB462 (luxdegalutamide) in combination with abiraterone in adult male patients with metastatic hormone-sensitive prostate cancer (mHSPC)

mHSPC

mCRPC
- An Open-Label, Phase 1/1b Study of ORIC-944 as a Single Agent or in Combination with an Androgen Receptor Pathway Inhibitor in Patients with Metastatic Prostate Cancer
- A Multinational, Multicenter, Prospective, Randomized, Controlled, Open-Label, Phase 2/3 Study of Lutetium Rosopatamab Tetraxetan plus Standard of Care vs. Standard of Care alone for Patients with PSMA Positive Metastatic Castration-Resistant Prostate Cancer Previously Treated with Androgen Receptor Pathway Inhibitors
- A Phase 3 Randomized, Double-blind, Placebo-controlled Study of Pasritamig (JNJ-78278343), a T-cell-redirecting Agent Targeting Human Kallikrein 2, + Best SupportiveCare Versus Best Supportive Care for Metastatic Castration-resistant Prostate Cancer
- A Phase 3 Randomized, Open-label Study of Pasritamig (JNJ-78278343), a T-cell-redirecting Agent Targeting Human Kallikrein 2, With Docetaxel Versus Docetaxel for Metastatic Castration-resistant Prostate Cancer
- A phase 3 study of MK-5684 versus alternative NHA in mCRPC
Inclusion Criteria
- Potential subjects are men aged ≥18 years who are clinically indicated for treatment with Provenge (asymptomatic or minimally symptomatic metastatic castrate-resistant [hormone refractory] prostate cancer)
- Have qualified for on-label Provenge infusion
- Have received all 3 infusions of Provenge prior to randomization
- Written informed consent provided prior to the initiation of study procedures
- Estimated life expectancy ≥12 months
Exclusion Criteria
- Men who are not clinically indicated for treatment with Provenge (asymptomatic or minimally symptomatic metastatic castrate-resistant [hormone refractory] prostate cancer)
- Need for systemic chronic immunosuppressive therapy (eg, antitumor necrosis factor alpha monoclonal antibodies, or glucocorticoids)
- Uncontrolled, concurrent illness including, but not limited to the following: ongoing or active infection (bacterial, viral, or fungal), or psychiatric illness that would limit compliance with study requirements, as well as any condition that would preclude a subject from completing Provenge or sipuleucel-T treatment
- On experimental or investigational therapy